COVID-19 News: Cue OTC test, EUA Public FAERS dashboard, Roche false results, Ivermectin warning, Learn about treatment options
CUE Test for Home and Over The Counter (OTC) Use
Cue Health
INDICATION FOR USE: Qualitative detection of nucleic acid from SARS-CoV-2 in anterior nasal (nasal) swab specimens collected with the Cue Sample Wand.
Intended for use in adults (self-swabbing) or children ≥2 years of age (swabbed by an adult) with or without symptoms or other epidemiological reasons to suspect COVID-19. For Home and OTC use
authorized for non-prescription home use.
ADDRESSING UNMET NEED: First molecular OTC diagnostic test to continue to expand access to testing to help us in the fight against the pandemic
DESCRIPTION:
- For qualitative detection of nucleic acid from SARS-CoV-2 in anterior nasal (nasal) swab specimens collected with the Cue Sample Wand
- Test includes: the single-use Cue COVID-19 Test Cartridge, the single-use Cue Sample Wand nasal swab, the Cue Cartridge Reader the Cue Health Mobile Application (App)
- Battery-operated Cue Cartridge Reader runs the Cue Test Cartridge and communicates results directly to the Cue Health App in about 20 minutes
SENSITIVITY and SPECIFICITY:
- Correctly identified 96% of positive samples from individuals known to have symptoms
- Correctly identified 100% of positive samples from individuals without symptoms
REGULATORY PATHWAY: EUA
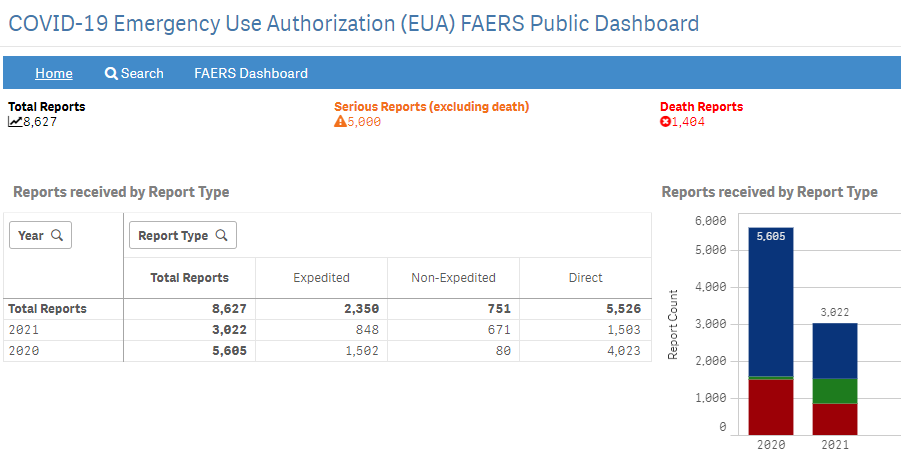
New COVID-19 EUA FAERS Public Dashboard
Launch of FDA Adverse Event Reporting System (FAERS) Public Dashboard to access data on adverse events
- User-friendly search tool for adverse events associated with the use of drugs and biologics
- Provides weekly updates of adverse event reports and increases transparency and provide more efficient access to safety information.
Potential for False Results with Roche Molecular Systems, Inc. cobas SARS-CoV-2 & Influenza Test
False positive results can occur with the Roche Molecular Systems, Inc. (Roche) cobas SARS-CoV-2 & Influenza A/B Nucleic Acid Test for use on the cobas Liat System
- Assay tubes may sporadically leak, causing an obstructed optical path in the Liat analyzer, producing abnormal PCR growth curves
- Abnormal PCR cycling in the reaction tubes may also produce abnormal PCR growth curves, leading to erroneous results
Recommendations
- Monitor for unexpected clusters of positive Flu B results
- Repeat tests when two or three analytes are positive
- Stop using the cobas Liat System and contact Roche if you suspect issues
Ivermectin should not be used treat or prevent COVID-19
Ivermectin has not been approved for treating or preventing COVID-19
- Approved to treat people with intestinal strongyloidiasis and onchocerciasis, two conditions caused by parasitic worms
- Different formulation used in animals to prevent heartworm disease and certain internal and external parasites
- FDA has not reviewed data to support use in COVID-19 patients; some initial research is underway
- Taking ivermectin is dangerous: Interaction with other medications e.g. blood-thinners, potential to overdose with nausea, vomiting, diarrhea, hypotension (low blood pressure), allergic reactions (itching and hives), dizziness, ataxia (problems with balance), seizures, coma and even death

Learn about treatment options available today
Image credit: Cue, Roche, FDA