News & Views: Flu vaccine supply, Rare Disease Day, Childhood Cancer Advocacy Forum, Heart Devices, Physiologically Based Pharmacokinetic (PBPK) Modeling
 Ensuring Supply of Influenza Vaccine
Ensuring Supply of Influenza Vaccine
Starts well before the current flu season ends – a year-round initiative
- Vaccine composition changes every year as flu viruses are constantly evolving
- Collaboration of FDA, WHO, CDC to evaluate global circulating strains and disease trends
- FDA advisory committee recommend 3-4 strains to include in trivalent and quadrivalent influenza vaccines
- Manufacturers begin manufacturing process to include the newly selected flu strains and seek FDA approval for lot release – quality control tests, including testing for sterility
 Observation of Rare Disease Day
Observation of Rare Disease Day
Created to raise awareness about the 7,000 known rare diseases with theme of Bridging Health and Social Care
- public meeting – “Patient Perspectives of the Impact of Rare Diseases: Bridging the Commonalities” to obtain perspectives on impacts on daily life
- latest FDA Voices post, on progress made and continued patient engagement efforts
- video on Patients Matter: Giving Patients A Seat at the Table– ro share their unique perspectives, experiences, and concerns
 Oncology Center of Excellence Childhood Cancer Advocacy Forum
Oncology Center of Excellence Childhood Cancer Advocacy Forum
Topics for Discussion:
- FDA’s External Engagement and Patient Advocacy: A Dialogue to Better Inform FDA of Patient Needs and Priorities
- Avoiding Pitfalls in Drug Development
- Approved Cancer Drugs in Children
- BPCA/WR Study Results
- Expanded Access to Investigational Drugs
- FDARA Implementation
- Pediatric PROs: Are they feasible?
Register here for in-person or online attendance
 Heart Devices regulated by FDA
Heart Devices regulated by FDA
- Automated external defibrillators (AEDs): Portable and automatic, to restore normal heart rhythm
- Cardiac ablation catheters: Long, thin flexible tubes. to treat abnormally rapid heartbeats
- Cardiovascular angioplasty devices: Long, thin, flexible tubes threaded into heart vessel to open narrowed or blocked areas
- Cardiac pacemakers: Small, battery-powered, implanted permanently, monitor heart’s electrical impulses. deliver electrical stimulation if bradycardia
- Implantable cardioverter defibrillators (ICDs): Monitor heart rhythms and deliver shocks if dangerous tachycardia
- Prosthetic (artificial) heart valves: Replacing diseased or dysfunctional heart valves,
- Stents: Small, lattice-shaped, metal tubes, some with drugs, to improve blood flow
- Ventricular assist devices (VADs): Mechanical pumps for short-term or long term use
Warning signs and symptoms of a heart attack
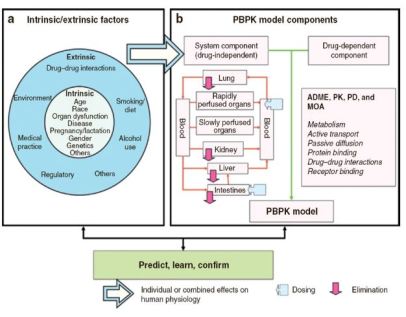 Supporting Drug Development Through Physiologically Based Pharmacokinetic (PBPK) Modeling
Supporting Drug Development Through Physiologically Based Pharmacokinetic (PBPK) Modeling
PBPK Models to Mechanistically Predict Drug Pharmacokinetics
- Grounded in human physiology – describe time course of absorption, distribution, metabolism, and elimination of one or more drugs
- Includes anatomical or physiological parameters such as blood flow, tissue composition, enzyme abundance, drug-specific values, such as tissue-to-plasma concentration ratios, metabolism and clearance
- Make predictions on drug concentrations achieved in the absence of clinical data
- Focus to improve accuracy of predictions and incoporate PBPK models in drug development
image credit: FDA