

INDICATION FOR USE: Maintenance treatment of adult patients with recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer who are in a complete or partial response to platinum-based chemotherapy
- Complementary diagnostic test, FoundationFocusTM CDx BRCA LOH, for tumor samples to determine homologous recombination deficiency (HRD) status.
EFFICACY:
- Randomized, double-blind, n=561, with recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer, RUCAPARIB vs. placebo
- Tumor tissue samples examined with next-generation sequencing assay for deleterious somatic or germline BRCA mutation (tBRCA); also percentage of genomic loss of heterozygosity (LOH); positive (HRD) status was tBRCA-positive and/or LOH high
- Median progression-free survival (PFS): 10.8 vs. 5.4 months, p<0.0001, 13.6 vs. 5.4 months (HRD, p<0,0001), 16.6 vs 5.4 months (tBRCA, p<0.0001)
SAFETY:
- Most common adverse reactions: Nausea, fatigue (including asthenia), abdominal pain/distension, rash, dysgeusia, anemia, ALT/AST elevation, constipation, vomiting, diarrhea, thrombocytopenia, nasopharyngitis/URI, stomatitis, decreased appetite, and neutropenia
- Myelodysplastic syndrome and/or acute myeloid leukemia
REGULATORY PATHWAY: sNDA
- Priority Review, completes initial accelerated approval commitments
- Initial approval for deleterious BRCA mutation (germline and/or somatic)-associated epithelial ovarian, fallopian tube, or primary peritoneal cancer

TAFINLAR (dabrafenib) and MEKINIST (trametinib)
Novartis
INDICATION: TAFINLAR is indicated, in combination with trametinib, for the treatment of patients with locally advanced or metastatic anaplastic thyroid cancer (ATC) with BRAF V600E mutation and with no satisfactory locoregional treatment options
ADDRESSING UNMET NEED:
- Anaplastic thyroid cancer (ATC) is rare, aggressive; accounts for about 1 to 2 percent of all thyroid cancers
- First FDA-approved treatment for patients with specific gene mutation
- Targeting same molecular pathway in diverse diseases is an effective way to expedite the development of treatments that may help more patients
MECHANISM OF ACTION:
- Dabrafenib and trametinib target two different kinases in the RAS/RAF/MEK/ERK pathway; used in combination resulted in greater growth inhibition of BRAF V600 mutation-positive tumor cell lines in vitro and prolonged inhibition of tumor growth in BRAF V600 mutation positive tumor xenografts compared with either drug alone.
EFFICACY:
- Open-label clinical trial, n=23, patients with rare cancers with the BRAF V600E mutation including ATC
- Partial reduction in tumore size: 57%
- Complete response: 4%
- No significant tumor growths for six months or longer: 64%
SAFETY:
- Side effects consistent with those seen in other cancers when the two drugs are used together
- Common side effect: Pyrexia, rash, chills, headache, arthralgia, cough, fatigue, nausea, vomiting, diarrhea, myalgia, dry skin, decreased appetite, edema, hemorrhage, hypertension, dyspnea
- Severe side effects of Tafinlar: Development of new cancers, growth of tumors in patients with BRAF wild-type tumors, serious bleeding problems, heart problems, severe eye problems, fever that may be severe, serious skin reactions, high blood sugar or worsening diabetes, and serious anemia.
- Severe side effects of Mekinist: Development of new cancers; serious bleeding problems; inflammation of intestines and perforation of the intestines; blood clots in the arms, legs or lungs; heart problems; severe eye problems; lung or breathing problems; fever that may be severe; serious skin reactions; and high blood sugar or worsening diabetes.
REGULATORY PATHWAY: sNDA
- Priority review, breakthrough therapy designation and orphan drug designation
- Both Tafinlar and Mekinist previously approved for use in BRAF V600 mutation-positive metastatic melanoma, BRAF V600E mutation-positive, metastatic non-small cell lung cancer
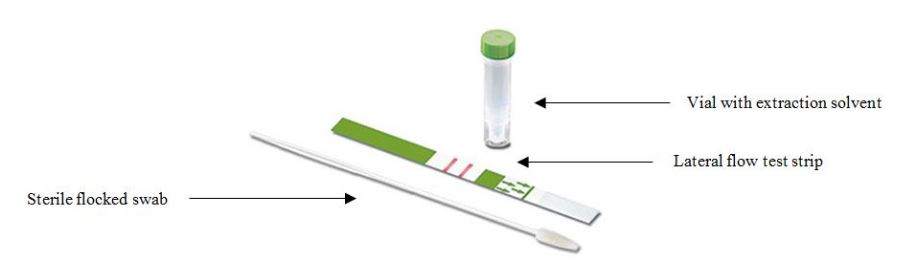
PartoSure test
Parsagen Diagnostics, Inc., Harvard Innovation Launch Lab
INDICATION FOR USE: Rapid, qualitative test for detecting the presence of placental alpha microglobulin 1 (PAMG-1) in cervicovaginal secretions.
Indicated as an aid to rapidly assess the risk of spontaneous preterm delivery in ≤ 7 days from the time of cervicovaginal sample collection in pregnant women with signs and symptoms of early preterm labor, intact amniotic membranes and minimal cervical dilatation (<3 cm), sampled between 24 weeks, 0 days and 34 weeks, 6 days gestation in women with a singleton gestation
ADDRESSING UNMET NEED:
- Spontaneous preterm delivery difficult to reliably predict
- This test aids assessment of spontaneous preterm delivery; improves upon
prediction of not undergoing preterm delivery
DESCRIPTION:
- Sterile, Vaginal Swab: Taking vaginal secretion
- Vial with Extraction Solvent: Saline solution with solubilizer and dispersant (Triton X100) and preservative (sodium azide); extracts vaginal secretions from swab
- Lateral Flow Test Strip: Lateral flow, immunochromatographic assay to identify presence PAMG-1; goat anti-mouse monoclonal antibodies at test region to detect PAMG-1 and goat anti-mouse anti-immunoglobulin antibodies at control region to detect IgG
ACCURACY AND PRECISION OF PREDICTION:
- For spontaneous preterm delivery in ≤ 7 days from the time of cervicovaginal sample collection in singleton pregnant women with signs and symptoms of early preterm labor, intact amniotic membranes and minimal cervical dilation (<3 cm)
- US Study: 6 preterm deliveries within 7 days from sample collection – 3 predicted and 3 missed, Sensitivity of 50%, Specificity of 98.4% (CI: 97.1% – 99.2%)
- Retrospective Spain study: 18 spontaneous preterm deliveries- 9 predicted, 9 were missed, Sensitivity of 50%, Specificity of 95.9%
RISK:
- False negative: Mother could progress unanticipated spontaneous preterm delivery without corticosteroids
- Increased risk or increased severity of respiratory distress syndrome, intracranial hemorrhage, necrotizing enterocolitis, and death compared with neonates whose mothers do receive antenatal corticosteroids.
REGULATORY PATHWAY: PMA
- Product Code: QBB
- Test results should always be considered in conjunction with other clinical evaluation and diagnostic procedures
- Post-approval studies required for additional data to verify safety and effectiveness

Cala ONE
Cala Health Inc.
INDICATION FOR USE: To aid in the transient relief of hand tremors in the treated hand following stimulation in adults with essential tremor
DESCRIPTION:
- Delivers individualized therapy that is calibrated by a physician using on-board sensors to measure the individual’s tremor
- Therapeutic device can be worn all day to provide on-demand
relief at home, in social settings, at work, or whenever patients desire relief.
GENERIC TYPE OF DEVICE: External upper limb tremor stimulator
- Prescription device which is placed externally on the upper limb and designed to aid in tremor symptom relief of the upper limb
EFFECTIVENESS AND SAFETY:
- Randomized, controlled, multi-center study
- Improvements in the treatment group in both physician and patient-rated measures of tremor severity compared to the sham group
RISKS AND MITIGATIONS:
- Tissue damage due to overstimulation: Non-clinical performance testing
Software verification, validation, and hazard analysis, Electrical safety testing
Shelf life testing, Labeling - Adverse tissue reaction: Biocompatibility evaluation, Labeling
- Electrical shock or burn: Electrical, thermal, and mechanical safety testing
Software verification, validation, and hazard analysis , Labeling - Interference with other devices: Electromagnetic compatibility (EMC) testing
Software verification, validation, and hazard analysis, Labeling
REGULATORY PATHWAY: De Novo request
- Regulation Number: 21 CFR 882.5897
- Regulation Name: External upper limb tremor stimulator
- Regulatory Class: Class II
- Product Code: QBC
Image Credits: Clovis, Novartis, Parsagen, Cala