FDA BRIEF: Week of February 5, 2018
Statement from Jeffrey Shuren, director CDRH: National Toxicology Program draft report on radiofrequency energy exposure
FDA ensures safety of electronic products that emit radiation (e.g. cell phones, TV)
- Federal Communications Commission (FCC) standards + other health agencies for scientific expertise
Recently released National Toxicology Program research on radiofrequency energy exposure in rodents
- Male rats showed carcinogenic activity with radiofrequency energy exposure rate much higher than current safety standard
- Equivocal/ambiguous evidence that whole body radiofrequency energy exposures in rats/mice actually caused cancer; additional unusual findings
- FDA to participate in peer review of study
- Public comment welcome Federal Register notice
FDA has reviewed many sources of scientific and medical evidence on possibility of adverse health effects from radiofrequency energy exposure
- Not found sufficient evidence of adverse health effects or increase in events like brain tumors
- Current safety limits for cell phones acceptable for protecting public health

CDER/CBER Data Standards Strategy FY2018-FY2022
Development, implementation, maintenance of comprehensive data standards program for pre- and postmarket regulatory review
- With Stakeholder collaboration
- Other Regulatory Authorities, Other Federal Agencies, Clinical Data Interchange Consortium (CDISC), Critical Path Institute, Health Level 7 (HL7), International Council on Harmonisation, International Organization for Standards, TransCelerate BioPharma, World Health Organization
Strategic Goals
- More efficient, science-based pre-market review
- Improve postmarket risk management strategies and pharmacovigilance
- Improve quality and integrity of marketed products
- Promote innovation in development and use of data standards
- Effective communication and collaboration with stakeholders
- Improve management and usability of information
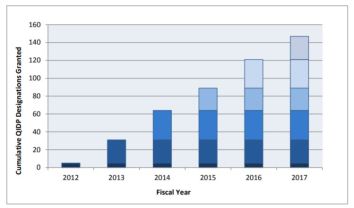
Generating Antibiotic Incentives Now (GAIN) FY2012-FY2017
GAIN passed in 2012 to address public health threat of antibacterial drug resistance by stimulating new antibacterial and antifungal drug development
- Qualified Infectious Disease Products (QIDP): Designation (147), Approval with Priority Review (12)
- Review and revision of guidance documents for streamlined development, limited population pathway for antibacterial and antifungal drugs (LPAD)
- Engagement to support development by public workshops and advisory committee meetings, collaborative partnerships, funding regulatory science research
- Development and implementation of stewardship programs to slow and control spread of resistant infections

FDA Helping Generic Industry Submit Complete Applications
Improve Abbreviated New Drug Application (ANDA) submission and assessment process;~ 50% ANDAs required at least 3 review cycles
- Guidance to industry
- Establishing internal practices to help reduce review cycles
Good ANDA Submission Practices to address
- Patent and Exclusivity Deficiencies
- Labeling Deficiencies
- Product Quality Deficiencies
- Bioequivalence Deficiencies

Statement from FDA Commissioner Scott Gottlieb: Scientific evidence on presence of opioid compounds in kratom, underscoring its potential for abuse
Scientific tools, data, research on FDA’s concerns about kratom’s potential for abuse, addiction, and serious health consequences; including death
- Public Health Assessment via Structural Evaluation (PHASE) methodology to use molecular structure to predict biological function
- Analyzed chemical structures of 25 most prevalent compounds in kratom
- All had structural similarities with controlled opioid analgesics, e.g. morphine derivatives
- Analyzed chemical structure against software to determine biologic targets
- 22 (including mitragynine) of 25 compounds bind to mu-opioid receptors
- Top five most prevalent compounds (including mitragynine) activate opioid receptors (“opioid agonists”)
- Some may bind to brain receptors and impact neurologic and cardiovascular functions – seizures and respiratory depression
- 3-D image on how strongly they bind to biological targets
- Comparable to scheduled opioid drugs
Learnings from reports of death associated with kratom
- Monitoring the use of kratom for several years, with import alerts
- Releasing reports of 36 deaths underscoring serious and deadly risks
- One new report of death with no known opioid use, except for kratom
Kratom should not be used to treat medical conditions, or as an alternative to prescription opioids
- No evidence of safety and effectiveness
