FDA BRIEF: Week of January 29, 2018
New steps to help prevent new addiction, curb abuse and overdose related to opioid products: OTC LOPERAMIDE
Unprecedented and novel action regarding OTC Loperamide (Brand Name Imodium A-D)
- Approved to help control short-term symptoms of diarrhea, including Travelers’ Diarrhea
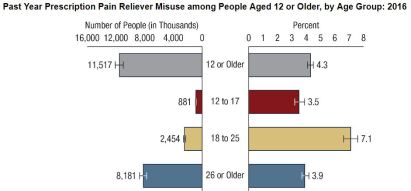
- However, intentional misuse and abuse of loperamide has been increasing; used as potential alternative to manage opioid withdrawal symptoms or to achieve euphoric effects of opioid use
- Reports of serious heart problems and deaths
Loperamide packaging limitations and unit-of-dose packaging
- Limited amount appropriate for use for short-term diarrhea per product label
- Manufactures to implement changes in a timely fashion
- Online retail web sites advised to take voluntary steps to limit distribution

Opioid REMS Education Blueprint
Blueprint for Extended-Release and Long-Acting Opioid Analgesic Risk Evaluation and Mitigation Strategy (ER/LA REMS) to ensure benefits outweigh risks
- Core educational messages for health care providers for pain
- Information on acute and chronic pain management, non-pharmacologic and pharmacologic treatments (non-opioid and opioid analgesic)
- Education for safe and effective use
Revised FDA Blueprint contains
- Core educational messages to be included in educational programs developed under Opioid Analgesic REMS
- Focus on fundamentals of acute and chronic pain management and contextual framework for safe prescribing
- Directed to prescribers, pharmacists, and nurses, but also relevant for other HCPs who participate in pain management of pain
 Payment and Reimbursement to Research Subjects
Payment and Reimbursement to Research Subjects
Updates to the Payment for Research Subjects: Information Sheet
- Reimbursement for travel expenses to and from the clinical trial site and associated costs such as airfare, parking, and lodging are acceptable
Image credit: SAMHSA, FDA