FDA Brief: Week of August 21, 2017
Early Feasibility Studies and Investigation Device Exemption
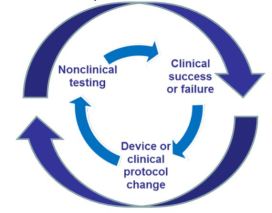
Early Feasibility Studies (EFS) Program provides opportunities to gain early clinical experience with innovative technology
- enrolls small number of subjects
- is used to evaluate the device design concept with respect to initial clinical safety and device functionality
- may guide device modifications
CDRH’s efforts to streamline the clinical trial initiative
- New EFS section added to Investigation Device Exemption(IDE) webpage in Device Advice
FDA is carefully evaluating prescription opioid medications approved to treat cough in children
Vital to understand potential complications in children using opioid-containing medications, even according to labeled instruction
- Added contraindication to drug labels alerting that codeine should not be used for any reason, including treatment of cough, in children younger than 12 years
Pediatric Advisory Committee scheduled on Sep 11th will focus on
- use of prescription opioid products containing hydrocodone or codeine for the treatment of cough in pediatric patients
- current treatment practices
- benefit-risk considerations
- help inform Agency’s decision-making processes related to these medications
Additional efforts:
- tips for consumers on how to safely treat a child’s cold
- funding research to develop comprehensive, consumer-centered approaches on best practices for the safe use of pediatric cough and cold medications
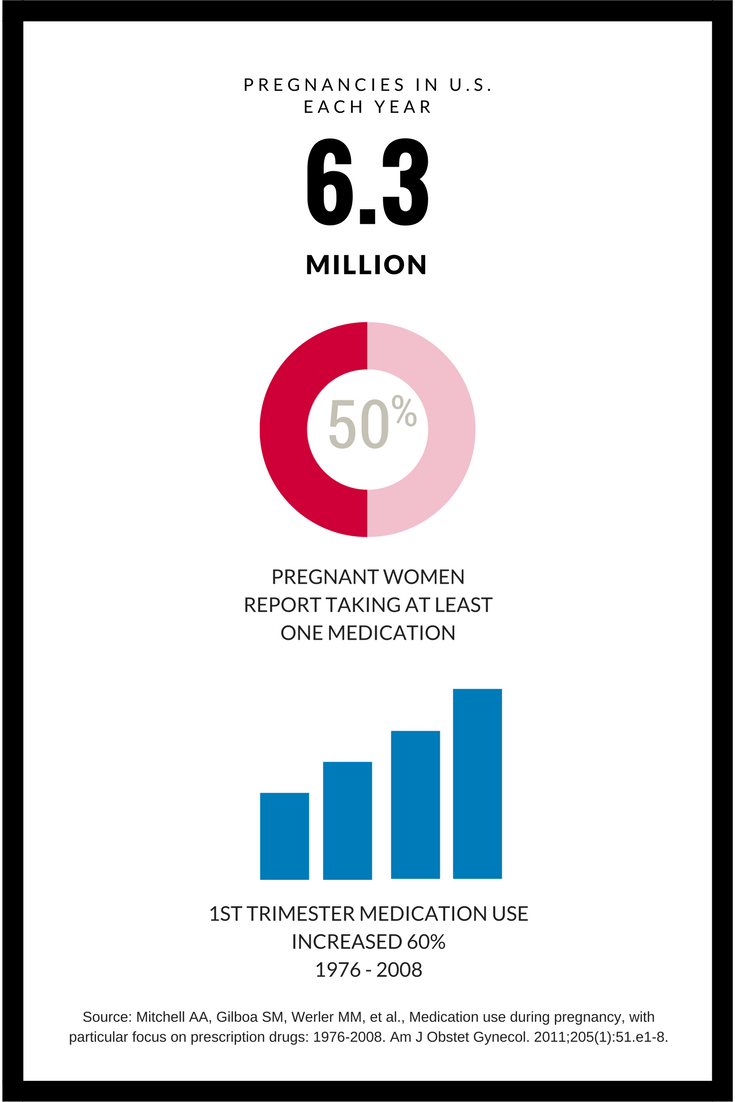
Working to Improve Information on Medication Use during Pregnancy
Very few prescription medications specifically approved for use during pregnancy
- About half of the 6.3 million preganant women/year take at least one medication
Task Force on Research Specific to Pregnant Women and Lactating Women (PRGLAC)
- Established by the 21st Century Cures Act
- Research aimed at optimizing therapies for pregnant women and nursing mothers
- Led by NIH’s Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD)
Labeling requirements for the pregnancy and lactation subsections
- Implemented in 2015
- Provided framework for clearly communicating benefits/risks
- Removed the decades-old pregnancy category letter system
Pregnancy research initiatives
- Led by Office of Women’s Health
- Using predictive modeling to anticipate response to drug
- Understand safety, efficacy and effects of products e.g. vaccine safety, MRI effects, drug toxicity
Medication Exposure in Pregnancy Risk Evaluation Program (MEPREP)
- Research collaboration between FDA, academia and health insurers
- Learn about medication effects by linking healthcare records for moms and babies
- Epidemiologic study to evaluate effect of prescription opioids
Real-World Research and Safety Monitoring
- Pregnancy Registry
- Pregnancy outreach activities
Image credit: FDA
.