FDA BRIEF: Week of March 13, 2017

 KISQALI (ribociclib) tablets
KISQALI (ribociclib) tablets
Novartis Pharmaceuticals Corporation, East Hanover, NJ, USA
INDICATION: In combination with an aromatase inhibitor as initial endocrine-based therapy for the treatment of postmenopausal women with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)- negative advanced or metastatic breast cancer.
UNMET NEED: Effective new treatment option for the continuing unmet need of the HR+/HER2- advanced breast cancer population.
REG PATHWAY: NDA
- Priority Review, Breakthrough Therapy Designation
- Post-marketing Requirements: Clinical trial to assess the efficacy and safety of an alternative dosing, clinical pharmacokinetic trial in severe renal impairment
MECHANISM OF ACTION: Inhibitor of cyclin-dependent kinase (CDK) 4 and 6 that are activated and play crucial role in cell cycle progression and cellular proliferation.
EFFICACY
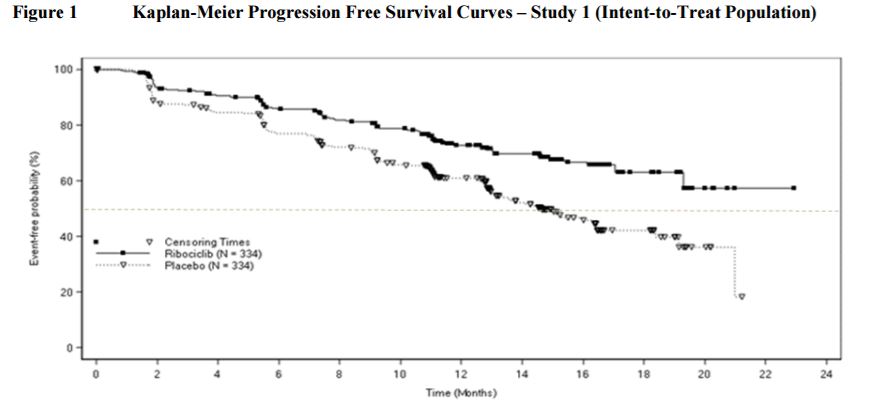
- Single randomized, double-blind, multicenter study (n=668), postmenopausal women with HR-positive, HER2-negative, advanced breast cancer with no prior therap, 21 days, 7 days off, KISQALI plus letrozole vs. placebo plus letrozole
- Primary Endpoint: Investigator-assessed progression-free survival (PFS) using Response Evaluation Criteria in Solid Tumors (RECIST) v1.1.
- Pre-planned interim analysis PFS: 27.8% vs. 44.9%, p< 0.0001
SAFETY:
- Most common adverse reactions: Neutropenia, nausea, fatigue, diarrhea, leukopenia, alopecia, vomiting, constipation, headache, and back pain
- Most common grade 3 or 4 adverse reactions: Neutropenia, leukopenia, abnormal liver function tests, lymphopenia, and vomiting
- Warning and Precaution: Prolongation of QT interval

KEYTRUDA (pembrolizumab) injection
SUPPLEMENTARY INDICATION: treatment of adult and pediatric patients with refractory classical Hodgkin lymphoma (cHL), or who have relapsed after 3 or more prior lines of therapy [see Clinical Studies (14.4)].
REG PATHWAY: Supplemental BLA
- Orphan Drug Designation, Breakthrough Therapy Designation, Priority Review, Accelerated Approval
- Accelerated approval based on tumor response rate and durability of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in the confirmatory trials.
EFFICACY:
- Single multicenter, non-randomized, open-label study, n=210, patients with relapsed or refractory cHL, KEYTRUDA every 3 weeks until unacceptable toxicity or documented disease progression, or for up to 24 months
- Major efficacy outcome measures: Overall Response Rate (ORR), Complete Response Rate (CRR), duration of response, assessed by blinded independent central review
- ORR 69% (95% CI: 62, 75); median follow-up 9.4 months
- Median response duration: 11.1 months
- Efficacy in pediatric patients extrapolated from results observed in adults.
SAFETY:
- Most common adverse reactions: Fatigue, pyrexia, cough, musculoskeletal pain, diarrhea, rash and hypertransaminasemia
- Additional common adverse reactions: Dyspnea, arthralgia, vomiting, nausea, pruritus, hypothyroidism, upper respiratory tract infections, headache, peripheral neuropathy, hyperbilirubinemia and increased creatinine
- Other immune-mediated adverse reactions: hyperthyroidism, pneumonitis, uveitis, myositis, myelitis and myocarditis
- Safety profile in the pediatric patients similar to adults