November 3, 2016

MANAGEMENT CONTROLS
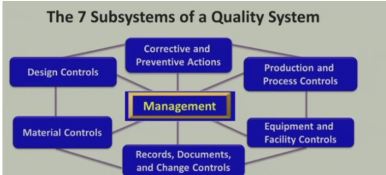
Management is Core – ultimately responsible for entire Quality System (QS)
- Involved, Engaged and Committed to the QS
- Senior Employee with executive responsibilities
- Harmonized with ISO and other global regulations
Implement Management Control Subsystem
- Adequate resources
- Ensure adequate and effective QS
- Monitor QS and make necessary adjustments
Management Responsibilities to Establish & Maintain
- Quality Policy
- Effective organization structure
- Appropriate responsibility and authority
- Adequate resources
- Appoint representatives
- Management reviews
- Establish Quality Plan
- Establish QS Procedures
Conduct Internal Audit
- Appropriate auditor, appropriate steps to address issues
- Appropriate documentation
Personnel
- With appropriate qualifications, training, background
