FDA Brief: Week of Feb 1, 2016


- Medical devices eligible for reimbursement labeled with an 11-digit reimbursement number – National Health Related Item Code (NHRIC) or National Drug Code (NDC) number
- The Unique Device Identification System final rule (UDI Rule) includes provision that rescinds any NHRIC or NDC number
- Draft guidance communicates
- Intent not to enforce before September 24, 2021
- Continued use of FDA labeler codes until September 24, 2018

- Guidance on appropriate human factors and usability engineering processes for safe and effective use – users, uses, use environment
- Improve device design to minimize potential use errors and resulting harm
- Risk Management by eliminating or reducing design-related problems
Guidance describes :
- Device Users, Use Environments and User Interface
- Preliminary Analyses and Evaluations
- Elimination or Reduction of Use-Related Hazards
- Human Factors Validation Testing
- Documentation
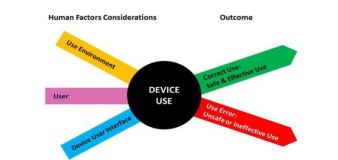

- Requirement of human factors data in premarket submissions based on potential for serious harm due to use error
- LISTED (for PMA, 510(k)) : Ablation generators, Anesthesia machines, Artificial pancreas systems, Auto injectors, Automated external defibrillators, Duodenoscopes, Gastroenterology-urology endoscopic ultrasound systems, Hemodialysis and peritoneal dialysis systems, Infusion pumps, Insulin delivery systems, Negative-pressure wound therapy, Robotic catheter manipulation systems, Robotic surgery devices, Ventilators, Ventricular assist devices
- NOT LISTED (for PMA, De Novo): Risk assessment on case-by-case basis; include criteria such as User interface modification, Different users, Recalls, adverse events, and problem reports, Device modifications